A baby’s life is saved with personalized gene therapy
Precision genetic medicine: from a case to a reproducible model
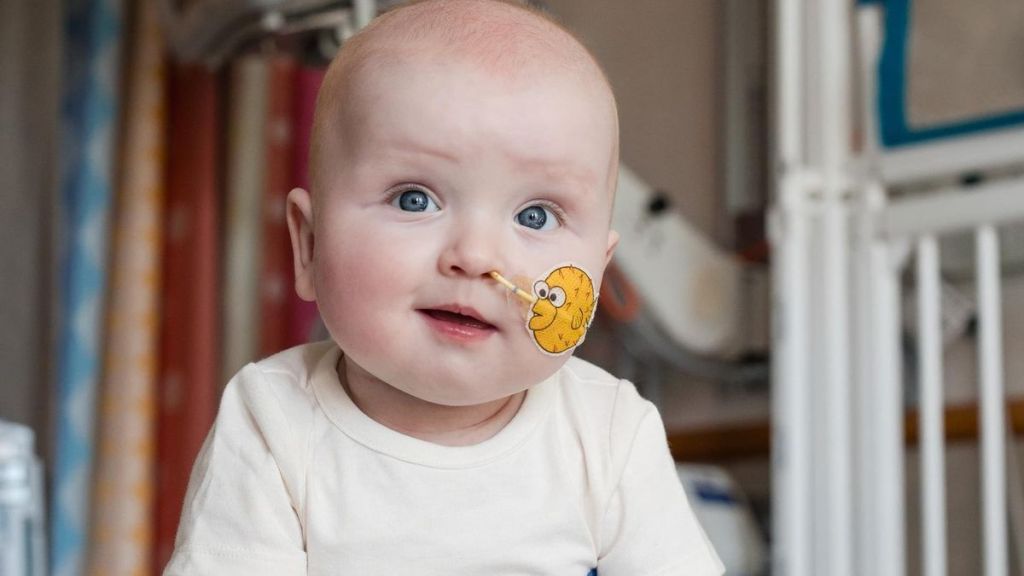
A group of researchers from different countries, in what can only be described as an unprecedented achievement, managed to develop, in just six months, a gene therapy for a single patient: a baby named KJ Muldoon, thus saving his life. This treatment was based on a technique called base editing and was able to correct the genetic mutation that caused a serious metabolic disease.
This case has opened up a new field in precision medicine, which consists of creating new personalized genetic therapies that could be effective in treating other rare diseases, currently incurable, and in record time.
The case that started it all
The CPS1 mutation KJ was born with prevents the production of a liver enzyme necessary to eliminate ammonia. The accumulation of this substance in the blood can lead to brain damage and even death.
Liver transplantation was, until now, the only known treatment, but it wasn’t always possible. However, Rebecca Ahrens-Nicklas of the Children’s Hospital of Philadelphia and Kiran Musunuru, a cardiologist at the University of Pennsylvania, implemented a new method: personalized gene therapy to correct the mutation that afflicted KJ.
To achieve this, they used the CRISPR-Cas9 gene editor in an advanced version called a base editor, and in this way, they were able to modify a single “letter” of the DNA in the defective gene. This small change was enough to restore the gene’s functionality and allowed KJ’s body to produce the CPS1 enzyme normally.
The effect has been amazing. KJ received the innovative therapy, and his ammonia levels decreased, while at the same time he developed normally, allowing his usual medication to be reduced. Currently, KJ is eating solid foods, learning to stand, and taking his first steps. His mother, Nicole Aaron, excitedly summarizes: “We celebrate every little milestone; he has a glow that lights up any room.”
The same strategy; other patients
The success achieved in KJ’s treatment has led many families, whose members suffer from rare diseases, to wonder if their opportunity will come soon.
The same health professionals who treated KJ are planning a clinical trial that will include between 5 and 15 participants affected by mutations that prevent the normal processing of ammonia.
The characteristic of this new work is its similarity to the previous one, since they will retain the same components of the base editor and only change the guide RNA, which directs it to the place where the specific mutation of each person is located.
This system will allow for other personalized treatments to be provided in less time (about 3 to 4 months) and at a lower cost. Furthermore, the U.S. Food and Drug Administration (FDA) has decided to validate, for new participants, the safety data corresponding to the gene therapy applied to KJ. This will reduce the number of trials.
New regulatory framework
Until now, the FDA required each new gene therapy proposal to undergo a lengthy regulatory process and a separate clinical trial, even if the differences compared to other treatments were minimal. Currently, with the advent of this type of personalized therapy, this requirement is under review.
For this reason, the researchers decided to publish their correspondence with the FDA. This way, other research teams developing similar treatments will have guidance to facilitate their work. “We want our work to be a tidal wave that lifts all boats,” commented Fyodor Urnov, one of the pioneers of gene editing at the Institute for Innovative Genomics at the University of California, Berkeley.
If this model moves forward, it could lead to a new era, with therapies designed on demand and according to each patient’s genetic characteristics, while also fitting into regulatory models that are expedited yet safe.
The heyday of personalized medicine
The increase in personalized therapy is not limited to the work that is the subject of this article, as several institutions have recently reported on similar projects.
The CRISPR Pediatric Cures Center, a collaboration between the University of California, Berkeley, and the University of California, San Francisco, aims to accelerate the development of personalized gene therapies. The U.S. government’s Health Advanced Research Projects Agency (HARPA) is also funding two programs focused on developing and implementing precision genetic medicine.
“I am more optimistic now than I was in the past,” says Joseph Hacia, a medical geneticist at the Keck School of Medicine of the University of Southern California in Los Angeles.
Challenges
Although the excitement is palpable, enormous challenges remain. Complex equipment, rigorous quality control processes, and high-security laboratories are needed. Implementing personalized therapy is extremely expensive.
Equity must also be considered so that everyone can access these treatments. Scientists are confident that as common components become standardized and regulatory frameworks are streamlined, costs will decrease and access will expand.
The role of biotechnology companies will be crucial in achieving sustainable business models that lead to personalized treatments, while maintaining their safety and efficacy.
A tailored therapy
Assuming the safety and efficacy of these studies are confirmed, and the FDA approves a treatment after testing it on only 10 or 15 participants, we would achieve what we could call a new class of expedited, personalized, and scalable gene therapies: we would enter a world unimaginable until now.
“Personalized treatments are definitely the direction we need to be heading,” says Ryan Maple, executive director of the Global Foundation for Peroxisomal Disorders in Tulsa, Oklahoma. He concludes, “This technology could be more than a game-changer. It could be revolutionary.”
The line between research and clinical practice can become very thin when, as in KJ’s case, urgency aligns with biotechnological innovation. Along this long path of science, we could say that certain biomedical advances are achieved letter by letter (of DNA).
Related

The End of Cultural Trenches: Why the Future of Faith Depends on a Weary Smile
Luis Herrera Campo
23 May, 2026
4 min

Randy Pausch’s Last Lecture: How to Play the Cards of Life
Marketing y Servicios
22 May, 2026
1 min

Responsibility for the Other and the Humanizing Power of Care
Observatorio de Bioética UCV
21 May, 2026
7 min

Can human suffering be measured?
Observatorio de Bioética UCV
21 May, 2026
4 min
 (EN)
(EN)
 (ES)
(ES)
 (IT)
(IT)

